Head of the laboratory
Gayane Ts. Gasparyan, Doctor of Chemical Sciences
ORСID: 0000-0002-2779-2728
Tel.: (+37410) 284-033
Email: gayanegas@gmail.com
The laboratory’s main research areas are:
- Production of tertiary phosphines, their oxides, and quaternary phosphonium salts
- Study of the nucleophilic and electrophilic addition reactions of quaternary phosphonium salts and the corresponding phosphine oxides, synthesis of new organophosphorus derivatives,
- Synthesis of diphenylphosphine oxides containing α,β-unsaturated groups, study of the reactions of their bromination and dehydrobromination products with nucleophiles A–H, where A = N, C, S, O, (alcohols, mercaptans, amines, CH-acids, azoles, etc.),
- Synthesis of new phosphoryl compounds containing a cyclopropane and aziridine ring in the radical
- Study of the biological properties of the synthesized compounds.
History of the Laboratory of Organophosphorus Compounds
The Laboratory of Organophosphorus Compounds was founded in 1970 by academician Maya. G. Indzhikyan of the National Academy of Sciences of the Republic of Armenia. From its inception, the Laboratory of Organophosphorus Compounds, under the direction of Academician M.G. Indzhikyan, began systematic research into the aqueous-alkaline cleavage of α,β- and β,γ-unsaturated quaternary phosphonium salts. This research, along with its theoretical interest, was of practical importance in terms of the possible synthesis of diphenylphosphoryl compounds with a specific structure, which could be of potential practical interest.
In 2014, the laboratory was headed by Professor Marlena Zh. Ovakimyan, Doctor of Chemical Sciences, who, expanding the scope of her research, continued research aimed at both identifying the specific features of the interaction reactions of quaternary phosphonium salts and the synthesis of new organophosphorus derivatives. During their years of leadership in the laboratory, M. G. Indzhikyan and M. Zh. Ovakimyan obtained many interesting results, including:
- A new class of compounds, phosphobetaines, which are 1:1 adducts of trialkylphosphines with various acetylene and allene compounds, was isolated and identified.
- An intramolecular dehydrogenation reaction of 2-hydrazino- and 2-hydroxylamino-ethylphosphonium salts and phosphonates was discovered, leading to phosphorus-substituted hydrazones and oximes.
- A new method for preparing 4-alkylsulfanyl-substituted 1,3-dienephosphonium salts, which are difficult to obtain by other methods, was proposed.
Since 2022, the laboratory has been headed by Gayane Ts. Gasparyan, a candidate of chemical sciences. While continuing her primary research focus, she has focused on identifying the specific reactions of 1,2-dibromoethyl- and α-bromovinyl-substituted phosphine oxides and phosphonates with various nucleophiles.
The laboratory’s work has resulted in the publication of numerous scientific papers and articles, and the receipt of patents.
Current Activities
The laboratory employs 10 highly qualified specialists, seven of whom hold PhDs in chemistry.
Best Results
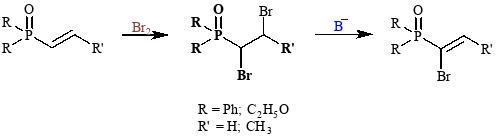
Laboratory staff synthesized α,β-unsaturated diphenylphosphine oxides and phosphonates, and conducted bromination and dehydrobromination reactions.
The resulting phosphorus-substituted mono- and brominated compounds were subjected to reactions with various nucleophiles to study their reaction patterns.
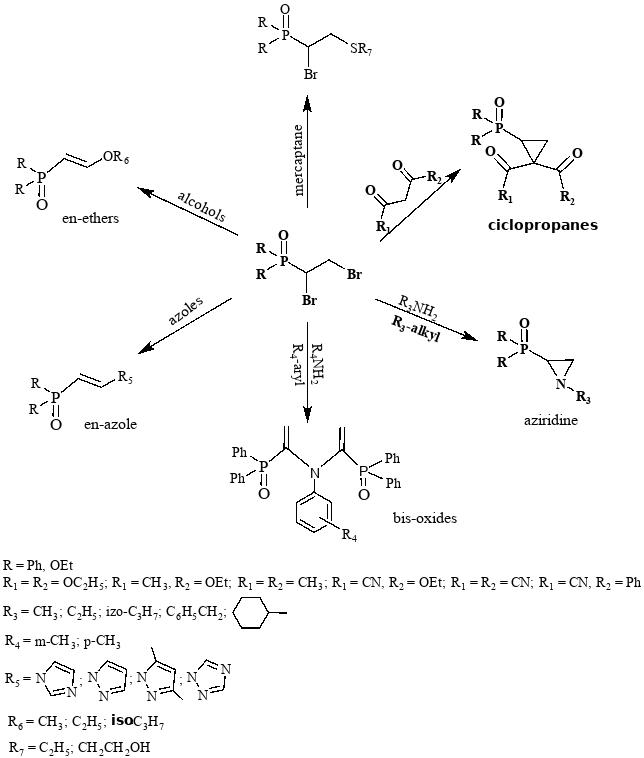
It was found that, depending on the nucleophile, in particular alcohols, CH-acids, primary alkyl and arylamines, mercaptans, and azoles, the interaction of bromo-substituted phosphine oxides and phosphonates leads to the formation of a wide range of phosphorus-substituted compounds, namely, en-ethers, cyclopropanes, aziridines, bisoxides, and en-azoles.
Based on the available literature data on the beneficial properties of organophosphorus compounds, it can be assumed that the synthesized compounds can also be used as medicinal preparations, insecticides, pesticides, plasticizers, stabilizers for polymeric materials, extractants for metal-containing salts, catalysts, etc.
The best works of recent years
Russ. J. Org. Chem., 2020, 56 (1), pp. 90–94. DOI: 10.1134/S1070428020010157
Russ. J. Gen. Chem., 2021, 91 (8), pp. 1494–1498. DOI: 10.1134/S1070363221080107
Russ. J. Org. Chem., 2022, 58 (12), pp. 1989–1991. DOI: 10.1134/S1070428022120314
Russ. J. Org. Chem., 2023, 59 (11), pp. 2008–2012. DOI: 10.1134/S1070428023110234
Russ. J. Gen. Chem., 2024, 94, (1), pp. 62-65. DOI: 10.1134/S1070363224010055
Russ. J. Gen. Chem., 2025, 95 (5), pp. 1086–1092. DOI: 10.1134/S1070363225600390
ЖОХ, 2025, 95 (7-8), с. 298-304. DOI: 10.31857/S0044460X25070062, EDN: SPTRKE
Russ. J. Org. Chem., 2025, 60 (12), pp. 2331–2335. DOI: 10.1134/S1070428024120030
Russ. J. Org. Chem., 2025, 61 (2), pp. 258–262. DOI: 10.1134/S1070428024603522